|
For example, 63Cu (29 protons and 34 neutrons) has a mass number of 63, and an isotopic mass in its nuclear ground state is 62.91367 u. For other isotopes, the isotopic mass usually differs and is usually within 0.1 u of the mass number. One unified atomic mass unit is approximately the mass of one nucleon (either a single proton or neutron) and is numerically equivalent to 1 g/mol.įor 12C, the atomic mass is exactly 12u, since the atomic mass unit is defined from it. One atomic mass unit is equal to 1.66 x 10 -24 grams. The unit of measure for mass is the atomic mass unit (amu). Therefore this resulting atomic mass is calculated from naturally-occurring isotopes and their abundance. Note that each element may contain more isotopes. How does the atomic number determine the chemical behavior of atoms? Atomic Mass of Chlorine Since the number of electrons is responsible for the chemical behavior of atoms, the atomic number identifies the various chemical elements. Chlorine is made in stars via the oxygen-burning and silicon-burning processes.Chlorine is a chemical element with atomic number 17 which means there are 17 protons and 17 electrons in the atomic structure. Walther Nernst explained the chain reaction mechanism for this phenomenon in 1918. 
In 1913, Max Bodenstein observed a mixture of chlorine gas and hydrogen gas exploded upon exposure to light. The first chain reaction to be discovered was a chemical reaction involving chlorine, not a nuclear reaction, as you might expect.Numerous radioactive isotopes of chlorine have been produced. Chlorine-35 accounts for 76% of the natural abundance of the element, with chlorine-37 making up the other 24% of the element. Natural chlorine consists of two stable isotopes: chlorine-35 and chlorine-37.These compounds are believed to have contributed significantly to the destruction of the ozone layer. While chlorine is still used in refrigerants, the number of chlorofluorocarbons (CFCs) released into the environment has dramatically declined. Chlorine compounds are used in medicines, plastics, antiseptics, insecticides, food, paint, solvents, and many other products. The compounds include chlorates, chloroform, synthetic rubber, carbon tetrachloride, and polyvinyl chloride. Chlorine is used to make drinking water safe, for bleaching, disinfection, textile processing, and to make numerous compounds. Pure chlorine is obtained primarily by electrolysis of saltwater.Soldiers could protect themselves from the gas by seeking higher ground and breathing through damp cloth since chlorine dissolves in water. The effectiveness of the gas was limited because its strong odor and distinctive color alerted troops to its presence. Later, the gas was also used by the Western Allies. The first use was in 1915 by the Germans in World War I. Because chlorine gas is toxic and because it's heavier than air, it was used as a chemical weapon.In particular, it's important to avoid mixing chlorine bleach with vinegar, ammonia, alcohol, or acetone. Since many household chemicals contain chlorine compounds, it's risky to mix them because toxic gases may be released. Exposure to 1 part per thousand in air may cause death. The gas irritates the respiratory system, skin, and eyes. While it's needed for survival, pure chlorine is extremely toxic. The element is usually obtained by eating salt, which is sodium chloride (NaCl). In the human body, it's found as the chloride ion, where it regulates osmotic pressure and pH and aids digestion in the stomach. Chlorine is essential for living organisms.There is so much chlorine in the Earth's oceans that it would weigh 5x more than our present atmosphere if it were somehow suddenly released as a gas.Chlorine is the 3rd most abundant element in the ocean (about 1.9% by mass) and 21st most abundant element in the Earth's crust.The Greek word chloros means greenish-yellow. Pure chlorine is a greenish-yellow gas or liquid with a distinctive odor (like chlorine bleach).It wasn't until 1811 that Sir Humphry Davy determined the gas was, in fact, a previously unidentified element. Scheele did not recognize this gas as a new element, instead believing it to contain oxygen. 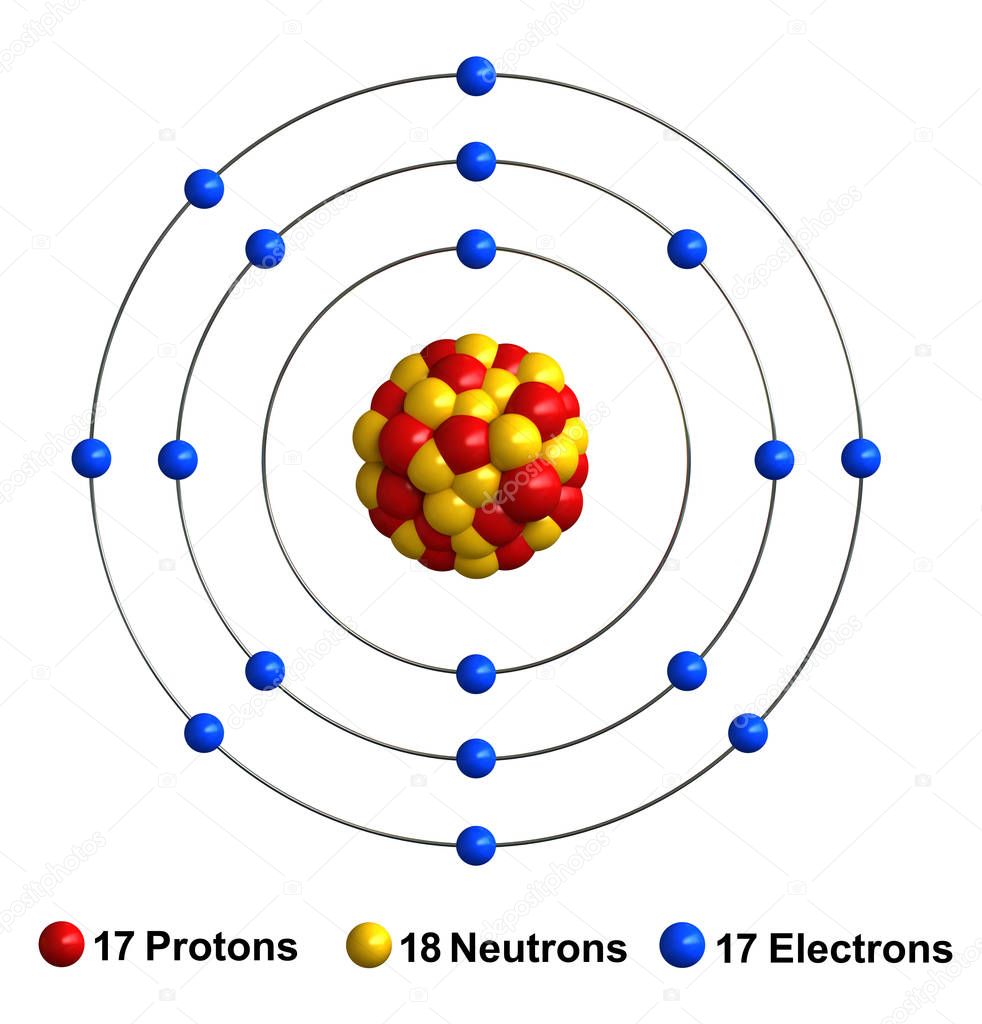
Although chlorine compounds have been used by man since ancient times, pure chlorine was not produced (on purpose) until 1774 when Carl Wilhelm Scheele reacted magnesium dioxide with spiritus salis (now known as hydrochloric acid) to form chlorine gas.Free chlorine is rare but exists as a dense, diatomic gas. Because of its high reactivity, chlorine is found in compounds. Like other halogens, it's an extremely reactive element that readily forms the -1 anion. It is the second lightest halogen, after fluorine. Chlorine belongs to the halogen element group.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
